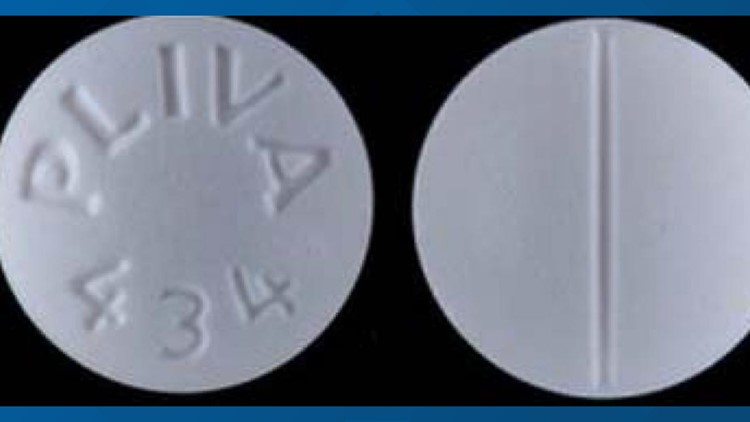
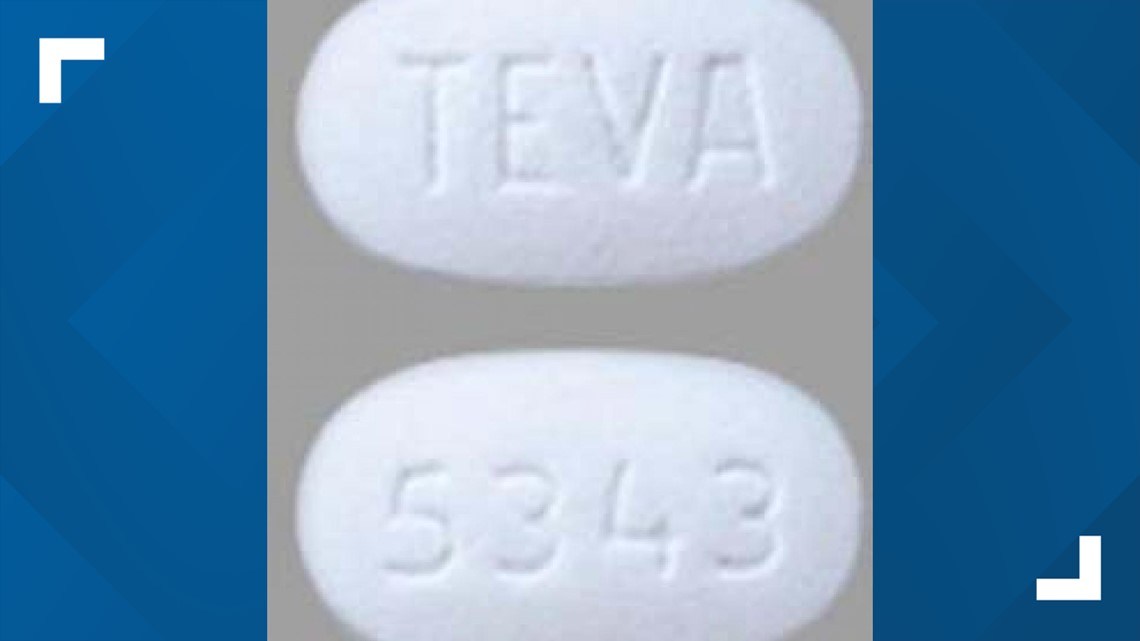
WASHINGTON — Pharmaceutical company AvKare has announced it is voluntarily recalling two lots of 100 mg tablets because a drug used to treat erectile dysfunction was mistakenly packaged together with an antidepressant.
A "product mix up" of Sildenafil and Trazodone caused the drugs to be "inadvertently packaged together during bottling at a 3rd party facility," the company said in a recall notice.
Sildenafil, the active ingredient in Viagra, is used for the treatment of erectile dysfunction. Trazodone is used to treat "major depressive disorder," according to the recall notice.
The company warned that unintended intake of Trazodone may result in adverse health consequences such as sedation, dizziness, constipation, and blurred vision.
Unintentional consumption of Sildenafil may pose serious health risks to those with underlying medical issues, such as lowering blood pressure to dangerous levels for those who are taking prescriptions containing nitrates.
The recalled bottles of Trazodone Hydrochloride are 1,000 count bottles from Lot 36783 with an expiration date of June 2022, NDC No. 42291-834-10.
The recalled bottles of Sildenafil are 100 count bottles from Lot 36884 with an expiration date of March 2022 and NDC No. 42291-748-01.

AvKare said the products were distributed nationwide by its distributors and wholesalers.
Consumers with questions regarding the recall can contact AvKARE at 1-855-361-3993 Monday- Friday (9am – 5pm EST).